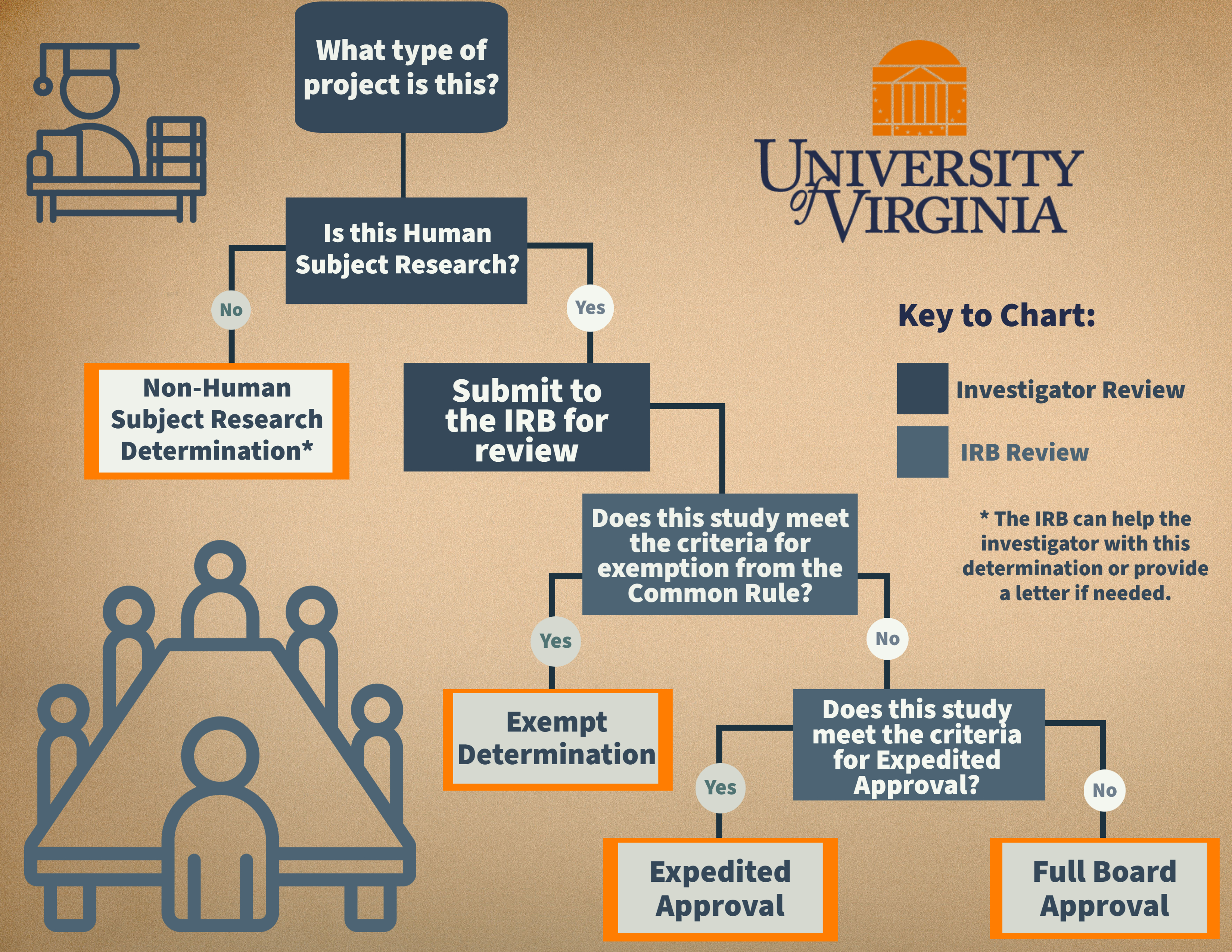
In order to ensure the rights, welfare, and protection of all subjects, all human subjects research regardless of sponsorship, must be reviewed and approved by the IRB prior to initiation. This includes all interventions and interactions with human subjects for research, including advertising, recruitment and/or screening of potential subjects. Before submitting a protocol, it is important to understand what categories of research fall under the IRB’s review and why. If you have further questions after reviewing the information below, send a summary of your project along with a copy of your grant application to the IRB-SBS Director: irbsbshelp@virginia.edu. We will review your email and determine if you will conduct human subject research and need IRB-SBS review.
The federal regulations define research as a systematic investigation including research development, testing, and evaluation, designed to develop or contribute to generalizable knowledge.
A "systematic investigation" is an activity that involves a prospective plan that incorporates data collection, either quantitative or qualitative, and data analysis to answer a question.
Examples of systematic investigations include (but are not limited to):
- surveys and questionnaires
- interviews and focus groups
- public or private observations
- analyses of existing data
- evaluations of social or educational programs
- cognitive and perceptual experiments
- medical chart (reviewed by IRB-HSR) or student record review studies
Investigations designed to develop or contribute to generalizable knowledge are those designed to:
- draw general conclusions,
- inform policy, or
- generalize findings beyond a single individual or an internal program (e.g., publications or presentations.)
However, research results do not have to be published or presented to qualify the experiment or data gathering as research. The intent to contribute to "generalizable (scholarly) knowledge" makes an experiment or data collection research, regardless of publication. Research that never is published is still research. Participants in research studies deserve protection whether or not the research is published.
Examples of activities that typically are not generalizable include:
- biographies
- oral histories that are designed solely to create a record of specific historical events
- service or course evaluations, unless they can be generalized to other individuals
- services, courses, or concepts where it is not the intention to share the results beyond the U.Va. community
- classroom exercises solely to fulfill course requirements or to train students in the use of particular methods or devices
- quality assurance activities designed to continuously improve the quality or performance of a department or program where it is not the intention to share the results beyond the U.Va. community.
Thesis or dissertation projects conducted to meet the requirement of a graduate degree are usually considered generalizable, and require IRB review and approval.
Human subjects research is any research or clinical investigation that involves human subjects. The federal regulations define a human subject as a living individual about whom an investigator conducting research obtains (1) data through intervention or interaction with the individual; or (2) identifiable private information.
Intervention includes both physical procedures by which data are gathered (e.g., survey) and manipulations of the subject or the subject's environment that are performed for research purposes. Interaction includes communication or interpersonal contact between investigator and subject. Private information includes information about behavior that occurs in a context in which an individual can reasonably expect that no observation or recording is taking place, and information which has been provided for specific purposes by an individual and which the individual can reasonably expect will not be made public (for example, a medical record). Private information must be individually identifiable (i.e., the identity of the subject is or may readily be ascertained by the investigator or associated with the information) in order for obtaining the information to constitute research involving human subjects.
Not all research that involves people is "human subjects research" but instead the individual is a "human source." The term “human source” defines situations where a researcher is interacting with another individual to gain knowledge about something but isn’t collecting personal information about that person. For example, the researcher wants to know the current policies for dealing with bullying at a school, so he calls a school administrator to ask that person about the school’s bullying policy and how it was developed, but does not ask about the administrator’s personal experience and how she felt about the policy. The school administrator is a human source but does not become a human subject until she provides personal information about her experience with the policy, bullying, etc. For more examples, please see "Examples".
Preliminary study activities, class projects, and other exploratory activities can be a gray area as to whether they need IRB approval. For example, if you have an assignment for your research methods class to conduct a survey, compile the results, and submit the paper for a grade, this activity does not satisfy the definition of research for an IRB review because the intent is not to generalize this information (instead the intent is to practice conducting research for a grade in a class). However, let’s suppose that you want to use this data for a thesis that will be published as part of your graduation requirement, which does meet the definition of research. Even though the initial collection did not fall under IRB review, because you want to use the data in a context that does require IRB review, the use of the data can be problematic. The federal regulations state that researchers cannot collect data before an IRB approves the data collection, except in the case where data was collected without the intent to conduct human subjects research. In this case, it can be argued that the data was initially collected without the intent to conduct human subjects research (at least according to the IRB definition of research), so the Board can allow its use. The catch here is that the Board will review the data collection process and will only approve the use of the data if the data were collected in an ethical manner. Going back to the survey example, if the surveys were collected in a manner that did not protect confidentiality, they may not allow you to use the data. Or if you conducted interviews, the Board may request that you seek the participants’ consent to use their data in the new study. In some cases, it may be easier to obtain IRB approval for the pre-study data collection than to try to navigate obtaining approval after the data is collected. If you think that your data collection will be a gray area or if you have already collected data, we recommend that you contact our staff for further assistance. They can help you determine if you should submit a protocol before you collect data and/or give you advice on how you can collect your data so that the Board can approve its use for a potential future study. For data that is already collected, the staff can help you to understand how that data can be used in a study.
Whether oral histories require IRB-SBS review or not depends on how the data will be used. If the purpose of gathering oral histories is to contribute to an archive, for example, the oral histories likely do not meet the requirements for IRB-SBS review as this is not considered a "systematic investigation" with the intent to generate "generalizable knowledge." However, if you intend to analyze the oral histories and draw conclusions from them, this is considered a "systematic investigation" with the intent to generate "generalizable knowledge." Often this distinction can be nuanced and we recommend that you contact our office for additional information. We can go through the details of your project and determine if you need to submit or not (and provide a letter indicating that you do not need to submit, if needed). For more information, see SOP 25.8.
Example of an Activity that Requires IRB Review |
Example of an Activity that Do Not Require IRB Review |
Differences Between Activities |
|---|---|---|
| Interviewing African American students about their experiences at various universities. | Interviewing a university official about admission procedures and programs. | The first example is collecting data from the individual that is about that individual ("human subject"). The second example does not collect personal information about the individual ("human source"). Interviewing the university official about his or her individual experience as an admissions counselor would require IRB review. |
| Conducting an anonymous survey to gather political opinions from middle class Republican voters for a thesis. | Conducting an anonymous survey at the end of a semester to gather information about the students’ perception of their instructor as part of an instructor improvement initiative. | The first example is collecting data for a generalizable event (i.e. thesis publication). The second example is for an internal evaluation only. If the evaluation data were to be used in a publication for an education research journal, this use of the data would require IRB review. |
| Analyzing student records to determine a correlation between test scores and free lunch status. |
Analyzing student records from students in the 1850s.
Studying interviews published in the library. |
Although both examples involved data that are already collected, the IRB is only concerned with sources of data that are the primary source and can be linked directly to a living individual. |