If your research plans have changed or you need to pause the review process, you can request that the IRB-SBS withdraw your protocol from review. Withdrawing a protocol will:
- Remove the protocol from the Pre-Reviewer and Board Reviewer’s assignment list
- Remove some of the review notes created by the Pre-Reviewer and Board reviewer
- Return the protocol to a state where it can be edited
- Allow you to resubmit the protocol at a later date
- Allow you to delete the protocol
Withdrawing a protocol will not:
- Delete the protocol
- Delete the comments made on the protocol from a previous review
If you know that a protocol cannot move forward or if it needs to be paused for more than a few months, please withdraw the protocol. Some of our reviewers’ time is spent checking on stale protocols and it will help our office efficiency by eliminating protocols from our lists. If you need to pause work on the protocol for only a month or two, it may be beneficial keeping it in the review cycle as some of the Pre-Reviewer and Board Reviewer work is deleted when a protocol is withdrawn. If you have questions about whether you should withdraw, contact your Pre-Reviewer or our office for assistance.
If after you withdraw a protocol, you want to remove the protocol from the Protocol Management page or permanently delete it, see Trash Bin/Recycle for more information.
Any protocol submitted to the IRB-SBS that has not been reviewed in a full board meeting (protocols in the pre-review phase of review and/or protocols that qualify for expedited or exempt review) can be withdrawn. This includes copies of the protocol that were created for a Modification review, Continuation review, and/or Unexpected Adverse Events review. Once a protocol is reviewed at a full board meeting, it cannot be withdrawn but the protocol version needs to be finalized according to the Board’s determination at the meeting. If the protocol version was "finalized" (approved or deferred at a board meeting) the protocol cannot be withdrawn; approved protocols can be closed. You will know that your protocol can be withdrawn if the “withdraw request form” link is active below the protocol name in the Protocol Management page. See “How to Withdraw an iProtocol” for more information. Requesting to withdraw a protocol can only be done by a Principal Investigator, Faculty Sponsor, Record Creator, or Contact Person.
- If the protocol is eligible for withdrawal, the “withdraw request form” link will be active below the protocol name in the Protocol Management page.
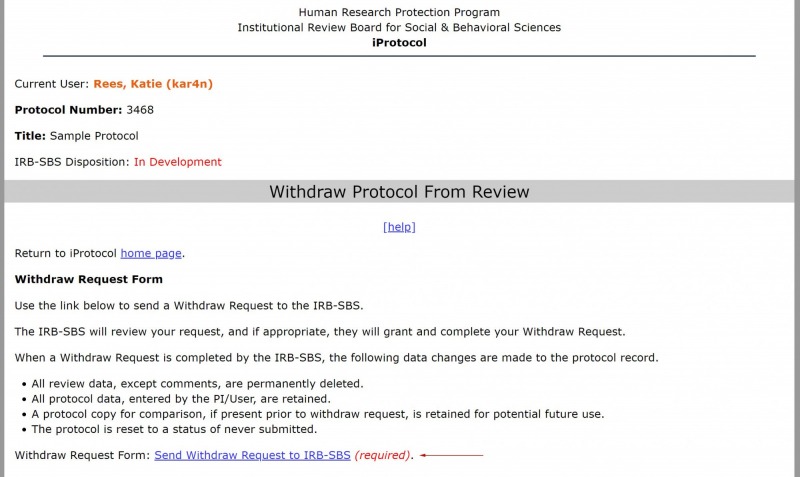
- Select the “withdraw request form” link.

3. Select the “Send Withdraw Request to IRB-SBS” link.
4. The “Success” message will display.
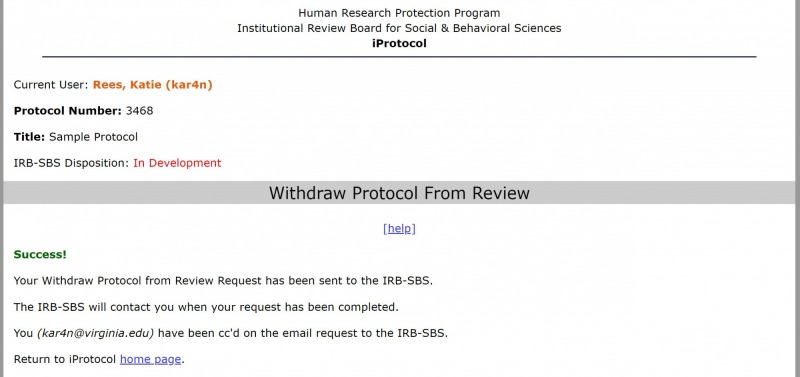
iProtocol will send a notification email to the Pre-Reviewer or Protocol Manager (if the protocol does not have a Pre-Reviewer assignment) who will complete the withdrawal process.
If the “withdraw request form” is still active, the withdrawal process has not been completed by the Pre-Reviewer or Protocol Manager. If you made the request in error, contact our office immediately so that they know to disregard the request.
Once the Pre-Reviewer or Protocol Manager completes the withdraw process, the Principal Investigator will be notified via email. The protocol listing in the Protocol Management page will denote that the protocol is withdrawn.

When the protocol is withdrawn, the protocol can be edited and resubmitted as needed.
If a copy is made of an approved protocol in order to submit a modification, continuation, unexpected adverse events report, or resubmit a deferred protocol for full board review, the copy can be withdrawn and the above rules and processes apply (copies made to create a new protocol are considered new protocols and the above rules and processes also apply). Once a copy of the protocol is withdrawn, the copy remains an editable version of the protocol. At this point you can opt to:
- Take no action: The copy of the protocol can remain in your Protocol Management page until such time as deemed necessary to edit and submit the version again. For example, if you needed to pause a modification review but want to revisit it in the future, you can request a withdrawal from review and then keep the copy to be updated and resubmitted at a later point. The “approved” version of the protocol remains the active version with the IRB-SBS and the copy is a working copy until it goes through the review process.
- Move the protocol copy to the Trash/Recycle Bin: The protocol version will be moved to the Trash/Recycle Bin so that it doesn’t clutter the Protocol Management page. The protocol copy is not permanently deleted and can be recovered to be used as an editable version of the protocol. Please note that you cannot make another copy of the protocol while there is an active copy of the protocol in the system. Instead, you need to recover the copy in the Trash/Recycle Bin and edit that version or use the "permanent delete" link to delete the protocol copy. See Trash/Recycle Bin for more information.
