2022 Ivy Biomedical Innovation Fund Overview
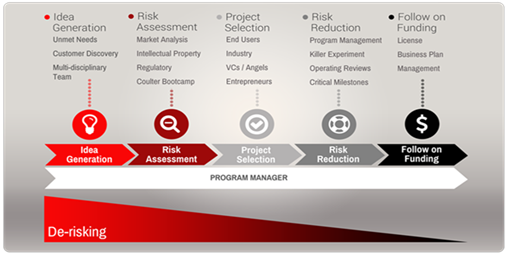
The Ivy Biomedical Innovation Fund was created by The Ivy Foundation to support biomedical innovation and translational research projects at the University of Virginia. The program plans to make 5 awards averaging $100K each for 12 months. The requested amount of funding must be commensurate with the project stage and goals. To generate and advance novel and compelling translational ideas, we strongly encourage projects that involve faculty co-investigators from multiple departments, schools, or specialties at UVA.
Program Objective:
The goal of this program is to support translational research projects that address unmet clinical needs and have a realistic path to delivering improvements in health care. Examples of desirable outcomes include: improved diagnosis and treatment of disease through new medical devices; new biomarkers or diagnostics; new therapeutic targets and agents; or new clinical adoption of developed tools. It is expected that outcomes from funded projects will result in new intellectual property, industry partnerships, license deals and/or the creation of a start-up companies.
Program Overview:
- Fund established in 2008
- Follow-on funding to advance funded projects: $21,815,063M
- 330 applications have been received to date
- 66 projects have been funded with 19 project renewals, 1 pilot project
- Average award size is $80,000 ($100K starting in 2020)
- Active patent applications: none in 2022
- Total transactions to date: 44 2022: 1 executed and 4 under CDA discussion
- Total Presentations & Publications: 153
- 10 presentations & 4 publications in 2022
Start-up companies launched catalyzed by after receiving Ivy Biomedical Innovation Funding: 9
2 companies currently in active discussions to launch in 2023
- PS Fertility (new in 2022)
- Advaray, Inc. (formerly BrachyFoam, LLC)
- EpiEP
- GenEp
- KeViRx
- RetiVue
- SoundPipe
- Sphynkx (no longer active)
- Tear Solutions